- light can be thought of as a stream of ``quanta'' called photons. Each
photon carries an energy
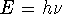 (h is ``Planck's constant'',
(h is ``Planck's constant'',  is the frequency).
is the frequency).
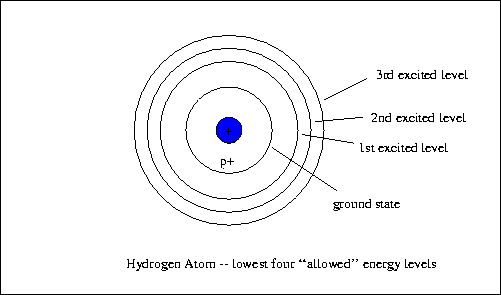
- Atoms had a crazy structure in which only certain orbits were allowed for the electrons - that word again, the orbits are ``quantized''.